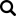Diagnostics and R&D
Diagnostics are the backbone of clinical medicine. Phibro offers a well-regarded diagnostics lab and the R&D capabilities necessary for autogenous vaccine manufacturing. We also have the ability to work closely with universities and state diagnostic laboratories to meet the autogenous vaccine needs of the industry in the U.S. Prompt, courteous, and accurate diagnostic service is critical to producing a quality product.
Our Services
• Precise bacterial isolation
• Serotyping of isolates
• Restriction Fragment Length Polymorphism (RFLP) – used to differentiate organism strains (DNA fingerprinting).
• Polymerase Chain Reaction (PCR) – used for identification and determination of the presence of specific virulence genes.
• Protein electrophoresis
• Western blotting
Offering Additional Value
A new bacterial growth procedure for Enhanced Antigen Surface Expression: “EASE™”. This laboratory technology creates nutrient restricted environments. This restrictive condition induces Gram-negative bacteria to hyper-express nutrient-capturing proteins on their surface. These proteins are highly antigenic and can be utilized by the animal’s immune system to strengthen its response against the pathogenic organisms.

Get the answers you need
Phibro Animal Health’s diagnostic and R&D professionals can answer many questions with existing in-house technology or can develop specific assays. Extensive efforts are focused on development and validation of new diagnostic tests, protein and antigen profiles, and customized growth and isolation media.
Track Your Orders From Start to Finish
User friendly, convenient tool that helps manage your Phibro autogenous vaccine orders 24/7 from your smartphone, tablet and/or computer:
- View isolates
- Check vaccine inventories
- Complete isolate extension paperwork
Visit VAC Tracker™ or download the app for Android or iOS
**Potency and efficacy of autogenous biologics have not been established.